Vaccine Market Trends: Immunization Programs, mRNA Technology & Forecast to 2034
Increasing government initiatives, growing awareness of preventive healthcare, and strong investments in biotechnology are accelerating expansion of the vaccine market worldwide.
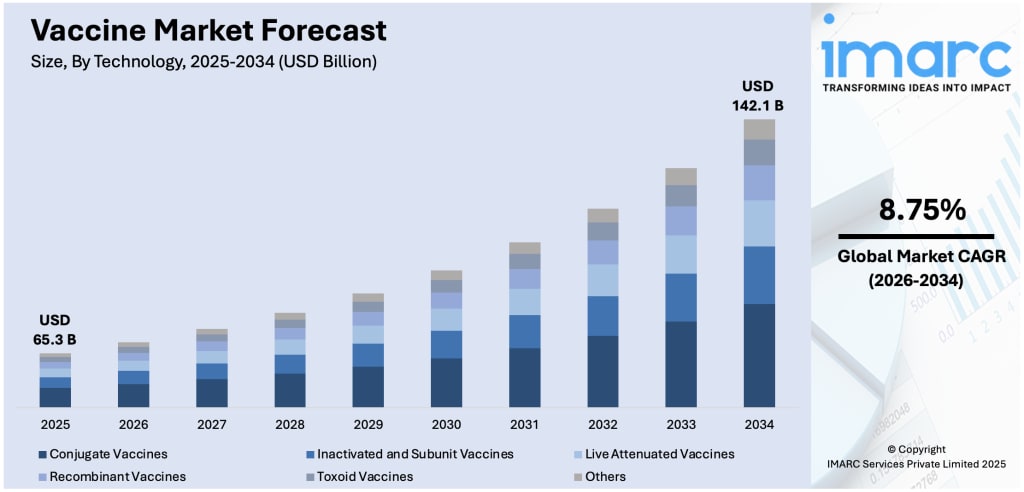
Rising infectious disease burden, breakthroughs in biotechnology, and an increasingly health-aware global population are reshaping how the world thinks about disease prevention. Vaccination has moved from a reactive emergency measure to a mainstream, long-term public health strategy — and the numbers tell that story clearly.
According to IMARC Group's latest research, the global vaccine market size was valued at USD 65.3 Billion in 2025. The market is projected to reach USD 142.1 Billion by 2034, at a CAGR of 8.75% during 2026-2034. Asia-Pacific currently leads the market with a dominant share of over 38.9% in 2025, driven by large-scale government immunization programs and rapidly expanding healthcare infrastructure across the region.
Report Attribute:- Key Statistics
Base Year:- 2025
Forecast Years:- 2026-2034
Historical Years:- 2020-2025
Market Size in 2025:- USD 65.3 Billion
Market Forecast in 2034:- USD 142.1 Billion
Market Growth Rate (2026-2034):- 8.75%
The market's trajectory is being shaped by a confluence of factors — from the roll-out of next-generation mRNA platforms to government mandates and growing adult immunization awareness. Pediatric vaccines continue to anchor demand, commanding around 56% of market share, while adult immunization programs are quickly catching up as healthcare systems expand booster campaigns globally.
Get a Sample Report for Actionable Market Insights
Vaccine Market Growth Drivers:
• Rising Incidence of Infectious Diseases
Diseases like seasonal influenza, HPV, hepatitis B, and emerging viral threats continue to push demand for effective immunization worldwide. Approximately 3,000 cases of bacterial meningitis occur annually in the United States alone, primarily affecting children and the elderly — a clear reminder of why preventive vaccination remains non-negotiable. The COVID-19 pandemic accelerated this urgency further, with surveys indicating that 63.7% of respondents globally now recognize vaccines as an effective tool against infectious disease spread. This heightened disease consciousness is directly translating into stronger uptake across both routine and emerging vaccine categories.
• Government Initiatives and Public Health Programs
National vaccination programs remain one of the most powerful demand engines in this market. The U.S. Vaccines for Children (VFC) program, for example, provides free vaccines to underserved pediatric populations, directly boosting coverage and market volume. In January 2025, China added Gardasil 9 to its subsidized immunization list, triggering the immediate production of 30 million additional doses. Meanwhile, the U.S. Department of Health and Human Services allocated USD 590 million to Moderna to expand mRNA capabilities and accelerate pandemic influenza vaccine development. These government-backed commitments are creating a reliable and expanding demand base that private players are actively building around.
• Technological Advances in Vaccine Development
The emergence of mRNA and recombinant vaccine platforms has fundamentally changed how quickly and effectively new vaccines can be developed. What once took years now takes months — Moderna's Nipah vaccine candidate entered Phase I human trials just nine months after a WHO outbreak alert, a timeline that was unthinkable a decade ago. Genomics and molecular biology tools allow scientists to identify vaccine candidates faster and with greater precision. The R&D investment backing this innovation is substantial: global vaccine R&D spending saw a 30% increase, reaching approximately USD 16 billion, following the COVID-19 vaccine breakthroughs that demonstrated mRNA's real-world scalability.
Vaccine Market Trends:
• Expansion of mRNA Platform Beyond COVID-19
mRNA technology, initially proven through COVID-19 vaccines, is rapidly expanding into new therapeutic territories. Moderna and Pfizer-BioNTech are actively developing mRNA-based flu vaccines, RSV shots, and even personalized cancer vaccines — with BioNTech initiating global trials for BNT116, targeting non-small cell lung cancer, in August 2024. In February 2025, Moderna partnered with OpenAI to use large language models for epitope prediction, compressing pre-clinical timelines from 18 months to just 6 months. This shift from single-disease tools to broad-platform technology is redefining vaccine development economics and competitive dynamics across the entire industry.
• Premiumization and Portfolio Diversification
Leading vaccine manufacturers are moving decisively beyond their core portfolios to capture emerging disease segments. GSK received FDA approval for Penmenvy, a meningococcal vaccine for individuals aged 10-25, in February 2025, while also expanding its RSV vaccine Arexvy to cover at-risk adults aged 18-49 — matching Pfizer and Moderna's earlier label expansions. Merck opened a new vaccine manufacturing facility in Durham, North Carolina in 2025, signaling long-term capacity confidence. The top four players — Pfizer, GSK, Sanofi, and Merck — collectively hold 70-75% of the market, but meaningful innovation is also coming from smaller biotech firms racing to cover niche but high-value indications.
• Growing Demand for Adult Immunization
The traditional focus on pediatric vaccines is broadening significantly as healthcare systems invest in adult immunization programs. RSV, shingles, and pneumococcal vaccines are seeing growing uptake among older adults, and the economics are compelling — Medicare faces approximately USD 3 billion in annual RSV hospitalization costs, making prevention a financially rational priority. GSK's shingles vaccine Shingrix earned approximately USD 4.30 billion globally, reflecting strong demand particularly in Europe and Australia. As awareness grows and reimbursement frameworks expand, adult immunization is becoming one of the fastest-growing and most commercially attractive segments in the global vaccine landscape.
Recent News and Developments in the Vaccine Market
• March 2025 — GSK Receives FDA Expansion for RSV Vaccine Arexvy
GSK secured FDA approval to expand Arexvy's use to at-risk adults aged 18-49, placing it on an equal footing with Pfizer's Abrysvo and Moderna's mResvia. The expansion reflects regulators' confidence in the vaccine's safety profile and addresses a significant unmet need for younger high-risk adults who were previously excluded from RSV immunization coverage.
• February 2025 — Moderna Partners with OpenAI for AI-Accelerated Vaccine Development
Moderna announced a strategic collaboration with OpenAI to leverage large language models for vaccine antigen and epitope prediction. The partnership has already demonstrated the ability to reduce pre-clinical development timelines from 18 months to approximately 6 months — a potentially transformative development for how the industry responds to both emerging outbreaks and planned vaccine pipeline builds.
• February 2025 — U.S. HHS Allocates USD 590 Million to Moderna for Pandemic mRNA Preparedness
The U.S. Department of Health and Human Services committed USD 590 million in funding to Moderna to strengthen mRNA manufacturing capabilities and fast-track the development of pandemic influenza vaccines. This investment reflects the U.S. government's long-term bet on mRNA as the foundational platform for future outbreak preparedness, ensuring supply chain resilience and production readiness.
• January 2025 — China Adds Gardasil 9 to National Subsidized Immunization Program
China's inclusion of Merck's HPV vaccine Gardasil 9 in its subsidized immunization list immediately triggered the production of 30 million additional doses. The move is expected to significantly expand HPV vaccination coverage in the world's most populous nation, creating substantial new market volume and reinforcing the broader trend of emerging markets institutionalizing vaccine access through government programs.
About the Creator
Rahul Pal
Market research professional with expertise in analyzing trends, consumer behavior, and market dynamics. Skilled in delivering actionable insights to support strategic decision-making and drive business growth across diverse industries.






Comments
There are no comments for this story
Be the first to respond and start the conversation.