Liquid Biopsy Market Trends: Cancer Diagnostics Innovation & Forecast to 2033
How advancements in non-invasive cancer diagnostics, biomarker detection, and genomic technologies are driving growth in the global liquid biopsy market
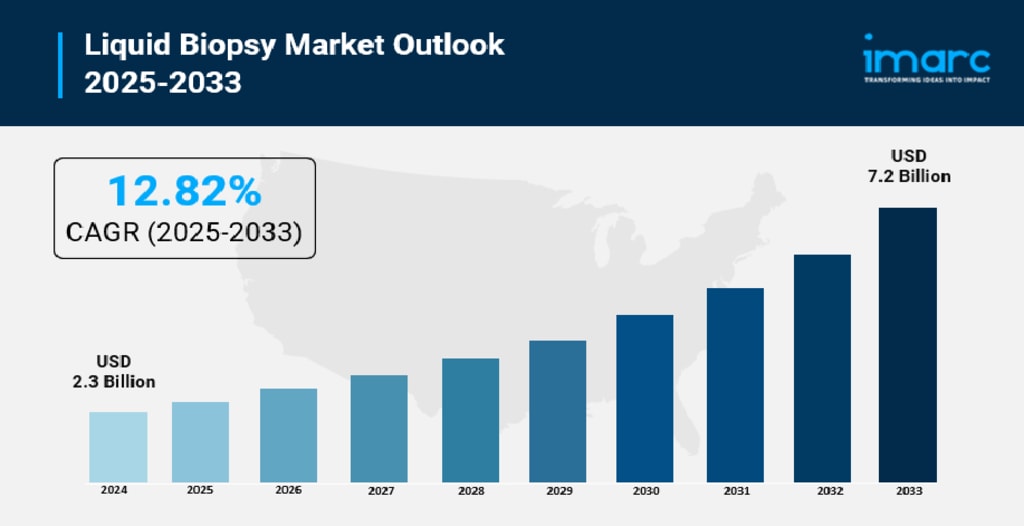
Rising cancer incidence globally, a strong push toward personalized oncology, and rapid innovation in non-invasive diagnostic platforms are together reshaping cancer care as we know it. According to IMARC Group’s latest research, the global liquid biopsy market size reached USD 2.3 Billion in 2024. Looking forward, IMARC Group expects the market to reach USD 7.2 Billion by 2033, exhibiting a CAGR of 12.82% during 2025-2033. North America currently leads the market, driven by advanced healthcare infrastructure, FDA-approved assays, and strong investment in precision medicine R&D.
Liquid biopsy is fundamentally changing how oncologists detect, monitor, and treat cancer. Rather than relying on painful tissue sampling procedures, clinicians can now draw a simple blood sample and analyze circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and exosomes to get a real-time snapshot of tumor activity. The WHO projected over 20 million new cancer cases globally in 2022 alone, and that number is expected to climb past 35 million by 2050 — making earlier, smarter, and less invasive diagnostics not just desirable but essential. Blood-based liquid biopsy tests now account for close to 87% of total market revenue, reflecting how quickly both clinicians and patients have embraced this approach. Key segments driving the market include kits and reagents, instruments and platforms, and testing services, with cancer detection and treatment monitoring representing the dominant application areas. Next-generation sequencing (NGS) leads the technology space, holding over 65% of market share in 2024, while North America continues to set the pace globally.
Evaluate Market Potential Before You Invest – Get the Report Sample
Liquid Biopsy Market Growth Drivers:
Surging Global Cancer Burden Driving Demand for Early Detection
With the WHO projecting more than 35 million new cancer cases annually by 2050 — up 77% from current levels — the pressure on healthcare systems to catch cancer earlier has never been greater. Liquid biopsy addresses this directly by detecting tumor-specific biomarkers like ctDNA and CTCs from a routine blood draw, enabling diagnosis at stages where treatment is far more effective. The American Cancer Society estimated over 2 million new cancer cases in the U.S. alone in 2025. This staggering disease burden is pushing hospitals, labs, and oncologists to integrate liquid biopsy into standard diagnostic workflows, fueling consistent market growth across all major regions.
Shift Toward Personalized Medicine and Targeted Therapies
Personalized oncology has moved from a niche concept to a clinical expectation, and liquid biopsy sits at the center of that shift. Tests like Guardant360 CDx and Foundation One Liquid CDx now allow oncologists to profile dozens of genomic alterations from a single blood sample, helping match patients to the right targeted therapies. In October 2024, Guardant Health received FDA approval for Guardant360 CDx as a companion diagnostic for EGFR mutations in non-small cell lung cancer — a milestone that signals growing regulatory confidence. As genomics becomes routine in treatment planning, the clinical utility and commercial demand for liquid biopsy are set to expand significantly.
Automated Platforms Lowering Barriers to Widespread Clinical Adoption
One of the biggest practical hurdles for liquid biopsy has always been throughput and turnaround time — but that’s changing fast. Automated sample processing systems reduced turnaround time by roughly 31% between 2023 and 2025, making it far easier for busy clinical labs to handle increasing test volumes. Thermo Fisher Scientific, for instance, introduced an automated extraction platform that cut processing time by 29%. More than 41% of laboratories have already transitioned from single-gene assays to comprehensive liquid biopsy panels. These operational improvements are lowering the technical barrier for smaller hospitals and independent labs, broadening the overall market base considerably.
Liquid Biopsy Market Trends:
Multi-Cancer Early Detection Tests Gaining Serious Clinical Traction
Multi-cancer early detection (MCED) tests are arguably the most exciting development in liquid biopsy right now. GRAIL’s Galleri test, which can screen for signals across more than 50 cancer types from a single blood draw, has been adopted by major healthcare providers in the U.S. and integrated into national early detection pilot programs. In June 2025, Guardant Health’s Shield MCD test received FDA Breakthrough Device designation for detecting bladder, colorectal, lung, and pancreatic cancers. These advances represent a genuine paradigm shift — moving cancer screening from a one-disease-at-a-time approach to broad, population-level detection with a simple blood test.
AI and NGS Integration Enhancing Diagnostic Accuracy and Speed
The combination of next-generation sequencing with artificial intelligence is rapidly elevating what liquid biopsy can deliver. NGS already commands over 76% of the liquid biopsy technology market, and integrating AI into result interpretation has reduced analysis errors by approximately 28% on current platforms. A January 2025 report in Bioinformatics Insights highlighted that AI-powered platforms can analyze complex genetic data from blood samples with high accuracy for real-time cancer monitoring. In April 2025, Illumina partnered with major oncology centers to embed NGS-based liquid biopsy panels directly into precision oncology workflows — a sign that AI-NGS integration is graduating from research to routine clinical practice.
Expanding Regulatory Approvals Accelerating Market Confidence
Regulatory momentum is a key tailwind for this market. The FDA has approved more than 14 liquid biopsy products in the U.S. to date, spanning cancer detection, therapy selection, and recurrence monitoring. In June 2025, Roche Diagnostics received FDA approval for a ctDNA-based test for therapy selection in non-small cell lung cancer. The NHS in England began offering liquid biopsy to women with mutated breast cancer in January 2025. These approvals aren’t just milestones — they directly enable reimbursement coverage, which is one of the most powerful drivers of physician adoption. As the regulatory and payer landscape matures, clinical uptake is accelerating across both established and emerging markets.
Recent News and Developments in Liquid Biopsy Market
• April 2025: Guardant Health announced FDA approval for an updated Guardant360 CDx liquid biopsy test, enabling comprehensive genomic profiling across all solid tumors from a single blood sample — a major expansion of its companion diagnostic reach.
• June 2025: Roche Diagnostics secured FDA approval for a ctDNA-based liquid biopsy test specifically designed for therapy selection in non-small cell lung cancer, reinforcing Roche’s position as a global leader in precision oncology diagnostics.
• June 2025: Guardant Health’s Shield multi-cancer detection test received FDA Breakthrough Device designation, marking a pivotal step toward mainstream adoption of pan-cancer blood-based screening for individuals aged 45 and older.
• April 2025: Labcorp expanded its precision oncology portfolio with two new offerings, including PGDx elio plasma focus Dx — the first and only FDA-authorized kitted liquid biopsy test for pan-solid tumors — broadening access to targeted therapy selection across cancer types.
• April 2025: Natera launched its ultra-sensitive Signatera Genome-MRD test in the U.S., advancing minimal residual disease monitoring with enhanced sensitivity that can detect tumor signals below 0.01% allele frequency — a significant leap for post-treatment surveillance.
Note: If you require specific details, data, or insights that are not currently included in the scope of this report, we are happy to accommodate your request. As part of our customization service, we will gather and provide the additional information you need, tailored to your specific requirements. Please let us know your exact needs, and we will ensure the report is updated accordingly to meet your expectations.
About the Creator
Suhaira Yusuf
I specialize in Consumer Insights, focusing on transforming detailed market data into strategic business solutions that accelerate growth and improve customer engagement.





Comments
There are no comments for this story
Be the first to respond and start the conversation.