HPV Testing Market Set for Strong Growth as Global Screening Efforts Intensify
Rising awareness, technological breakthroughs, and public health initiatives position HPV diagnostics as a cornerstone of cancer prevention worldwide
The global healthcare landscape is undergoing a quiet yet powerful transformation—one driven not by emergency interventions, but by prevention. At the center of this shift is Human Papillomavirus (HPV) testing, a diagnostic tool that is rapidly becoming indispensable in the fight against cervical and other HPV-related cancers. As awareness increases and screening programs expand across continents, the HPV testing market is witnessing remarkable growth.
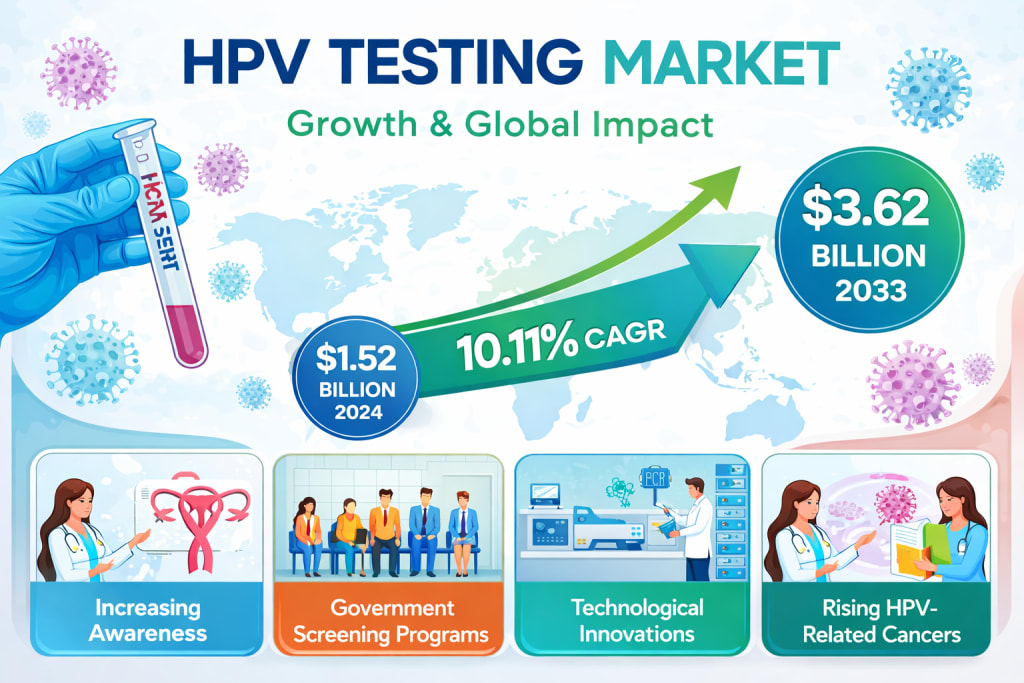
According to recent market insights, the Human Papillomavirus (HPV) Testing Market was valued at USD 1.52 billion in 2024 and is projected to reach USD 3.62 billion by 2033, growing at a compound annual growth rate (CAGR) of 10.11% from 2025 to 2033 . This trajectory underscores not only the rising demand for diagnostic solutions but also the global commitment to eliminating preventable cancers.
Understanding HPV Testing: A Preventive Breakthrough
HPV testing is designed to detect the presence of HPV DNA or RNA in cells, typically collected from the cervix. Unlike traditional screening methods that identify abnormal cells after they develop, HPV testing identifies high-risk viral infections before they cause cellular changes. This allows for earlier intervention, significantly improving patient outcomes.
HPV is one of the most common sexually transmitted infections worldwide. While many infections resolve naturally, persistent infections with high-risk HPV strains are known to cause cervical cancer, as well as cancers of the throat, anus, and other areas. The ability to detect these strains early has made HPV testing a cornerstone of modern preventive medicine.
Globally, healthcare systems are shifting from reactive treatment models to proactive screening approaches. HPV testing aligns perfectly with this paradigm, offering higher sensitivity and long-term risk prediction compared to traditional Pap smears.
Market Drivers: Awareness, Policy, and Innovation
One of the most significant drivers of the HPV testing market is the surge in awareness about cervical cancer prevention. Governments and international health organizations are actively promoting routine screening, particularly for women aged 30 and above. These initiatives are not only increasing testing rates but also normalizing conversations around sexual health and preventive care.
Large-scale public health campaigns have played a critical role in this transformation. Countries across North America, Europe, and Asia-Pacific are implementing national screening programs, often subsidized or fully funded. These efforts are complemented by global initiatives aimed at eliminating cervical cancer as a public health issue.
Technological innovation is another powerful growth catalyst. Advances in diagnostic technologies—such as polymerase chain reaction (PCR), next-generation sequencing, and liquid-based cytology—have significantly improved the accuracy and efficiency of HPV testing. Automated platforms and multiplex assays now allow laboratories to process large volumes of tests بسرعة and with greater precision.
Additionally, the emergence of self-sampling kits is revolutionizing accessibility. These kits enable women to collect samples in the privacy of their homes, addressing barriers such as stigma, lack of healthcare access, and discomfort with clinical procedures. This innovation is particularly impactful in low-resource settings, where traditional screening infrastructure may be limited.
Rising Disease Burden Fuels Demand
The increasing prevalence of HPV-related cancers is a major factor driving market growth. Cervical cancer remains one of the leading causes of cancer-related deaths among women globally. According to global health data, it ranks as the fourth most common cancer in women, with the vast majority of cases linked to high-risk HPV infections.
This growing disease burden has heightened the urgency for early detection strategies. Healthcare providers are increasingly recommending HPV testing as a primary screening tool, either alongside or in place of Pap tests. The shift reflects a broader recognition of HPV testing’s superior sensitivity and predictive value.
As more patients and clinicians prioritize preventive care, the demand for reliable, scalable diagnostic solutions continues to rise. This trend is expected to sustain long-term market growth.
Segment Insights: Technology and Application
Within the HPV testing market, cervical cancer screening remains the dominant application segment. Nearly all cases of cervical cancer are associated with persistent HPV infection, making early detection critical. Governments and healthcare organizations are actively promoting HPV-based screening as a more effective alternative to traditional methods.
From a technological standpoint, PCR-based testing leads the market. Known for its high sensitivity and specificity, PCR allows for the detection of multiple HPV genotypes in a single test. This capability enables accurate risk stratification and supports personalized patient management.
Other technologies, such as immunodiagnostics and hybrid capture, also play important roles. While immunodiagnostic tests are generally less sensitive than molecular methods, they offer cost-effective solutions for large-scale screening programs, particularly in resource-constrained environments.
The consumables segment—including reagents, assay kits, and sample collection devices—is another key contributor to market growth. With recurring testing and the rise of self-sampling kits, demand for high-quality consumables is increasing steadily, providing a consistent revenue stream for manufacturers.
Regional Landscape: Growth Across Continents
The HPV testing market exhibits strong regional variation, reflecting differences in healthcare infrastructure, policy frameworks, and awareness levels.
In the United States, the market is highly developed, supported by comprehensive screening guidelines and widespread insurance coverage. Advanced diagnostic technologies and strong regulatory backing have positioned the country as a leader in HPV testing innovation.
Europe is also experiencing steady growth, with countries like France integrating HPV testing into national screening programs. Government subsidies and robust public health systems are driving increased participation and test volumes.
In contrast, emerging markets such as India present significant growth potential. While adoption is currently concentrated in urban areas, government initiatives and NGO efforts are expanding access to low-cost testing solutions. Mobile health units and community outreach programs are helping bridge the gap between urban and rural healthcare.
The Middle East, particularly Saudi Arabia, is also witnessing growth as governments prioritize women’s health and preventive care. Awareness campaigns and healthcare reforms are gradually increasing acceptance of HPV testing in the region.
Challenges: Accessibility and Cost Barriers
Despite its promising outlook, the HPV testing market faces several challenges. One of the most pressing issues is limited access in low- and middle-income countries. In many regions, inadequate healthcare infrastructure, shortage of trained professionals, and lack of organized screening programs hinder widespread adoption.
Cultural stigma surrounding sexually transmitted infections further complicates the situation. In some communities, social taboos discourage women from seeking HPV testing, delaying diagnosis and treatment.
Cost is another significant barrier. Advanced diagnostic technologies, such as PCR and DNA sequencing, can be expensive to implement and maintain. For many healthcare systems, particularly in developing countries, these costs limit the scalability of screening programs.
Addressing these challenges will require a combination of policy interventions, technological innovation, and community engagement. Affordable point-of-care diagnostics, education campaigns, and international collaborations will be key to ensuring equitable access to HPV testing.
The Role of Innovation and Industry Players
The competitive landscape of the HPV testing market is shaped by leading diagnostic companies investing heavily in research and development. Innovations in assay design, automation, and data integration are enhancing test performance and operational efficiency.
Companies are also exploring partnerships with governments and non-profit organizations to expand access in underserved regions. These collaborations are crucial for scaling screening programs and achieving global health goals.
As the market evolves, the focus is shifting toward personalized diagnostics and integrated healthcare solutions. The integration of artificial intelligence and data analytics into diagnostic platforms holds the potential to further improve accuracy and streamline workflows.
Final Thoughts
The HPV testing market stands at a pivotal moment in its evolution. Driven by rising awareness, technological advancements, and global health initiatives, it is poised for sustained growth over the next decade. More importantly, it represents a critical tool in the fight against preventable cancers.
While challenges remain—particularly in terms of accessibility and affordability—the progress made so far is encouraging. With continued investment, innovation, and collaboration, HPV testing has the potential to transform women’s health outcomes worldwide.
As healthcare systems increasingly prioritize prevention over treatment, HPV testing will not only remain relevant but become essential. The journey toward eliminating cervical cancer is complex, but with tools like HPV diagnostics, it is no longer out of reach.





Comments
There are no comments for this story
Be the first to respond and start the conversation.