Global Cell and Gene Therapy Logistics Market Set for Rapid Expansion Through 2033
Rising Demand for Personalized Medicine Drives Transformation in Healthcare Supply Chains
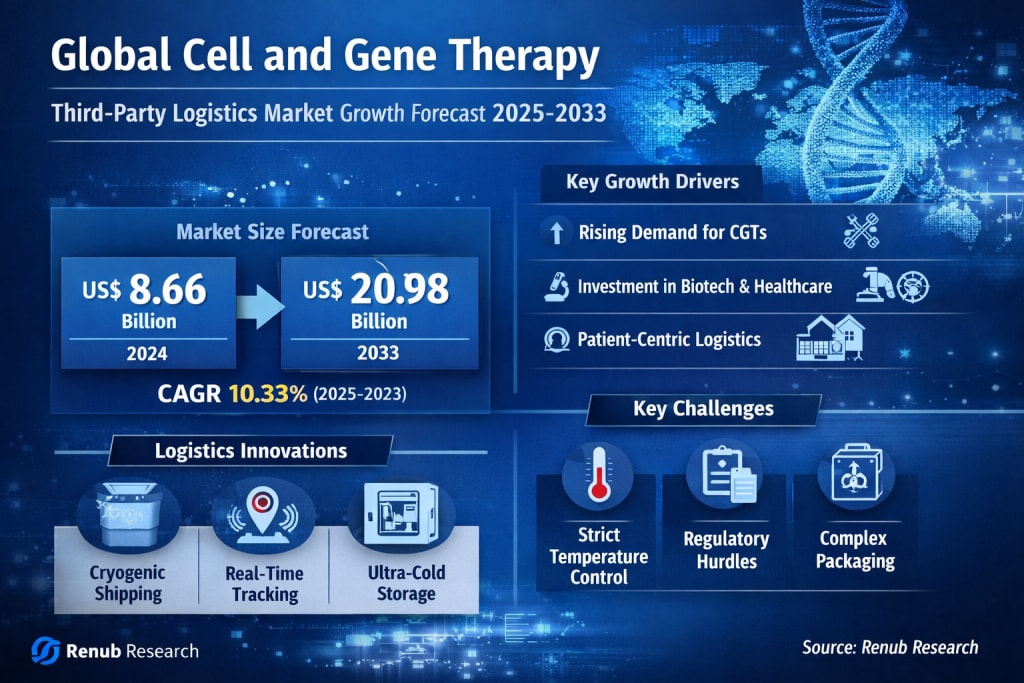
The global healthcare landscape is undergoing a profound transformation, fueled by the rise of personalized medicine and advanced therapeutic solutions. Among the most revolutionary developments are cell and gene therapies (CGTs), which are redefining how diseases—especially rare and chronic conditions—are treated. However, behind these cutting-edge treatments lies a highly specialized and rapidly evolving logistics ecosystem.
According to recent insights, the Global Cell and Gene Therapy Third-Party Logistics Market is projected to grow significantly, reaching US$ 20.98 billion by 2033 from US$ 8.66 billion in 2024, expanding at a compound annual growth rate (CAGR) of 10.33% from 2025 to 2033.
This surge is not just a reflection of increasing therapy approvals—it signals a fundamental shift in how healthcare products are stored, transported, and delivered to patients worldwide.
The Evolution of Logistics in the Age of Precision Medicine
Traditional pharmaceutical logistics systems were designed for mass-produced drugs with relatively stable storage requirements. In contrast, cell and gene therapies demand an entirely new level of precision. These treatments often involve living cells or genetically modified materials, which are extremely sensitive to environmental conditions.
As a result, logistics providers are now investing heavily in:
Ultra-low temperature storage systems
Cryogenic shipping solutions
Real-time tracking technologies
AI-powered supply chain optimization
These innovations are not optional—they are essential. Even minor deviations in temperature or handling conditions can compromise the integrity of a therapy, potentially rendering it ineffective or unsafe.
Why Demand for Specialized Logistics Is Surging
1. Explosion in Cell and Gene Therapy Development
One of the primary drivers of market growth is the increasing number of therapies moving from research to commercialization. Clinical trials for CGTs are expanding globally, requiring complex, multi-site logistics coordination.
Pharmaceutical and biotech companies are now conducting trials across continents, creating a need for:
Cross-border transportation
Regulatory compliance across jurisdictions
Time-sensitive delivery to trial sites
As more therapies receive regulatory approval, the demand for reliable logistics solutions continues to intensify.
2. Rise of Personalized and Patient-Centric Treatments
Unlike traditional drugs, many CGTs are customized for individual patients. This introduces a unique logistical challenge: delivering a one-of-a-kind therapy to a specific patient within a narrow time window.
This shift has given rise to patient-centric logistics, which includes:
Direct-to-patient delivery models
Real-time shipment visibility
Coordinated scheduling between hospitals and logistics providers
The goal is not just efficiency—but ensuring that life-saving treatments reach patients safely and on time.
3. Increased Investment in Biotechnology and Healthcare
Global investment in biotechnology is accelerating, with governments and private investors pouring resources into research and development. This growth is directly impacting logistics demand.
As new therapies emerge, companies are forming strategic partnerships with third-party logistics (3PL) providers to manage increasingly complex supply chains. These collaborations enable:
Faster market access
Scalable distribution networks
Enhanced compliance with regulatory standards
Technological Advancements Reshaping the Industry
Innovation is at the heart of the CGT logistics market. To meet the stringent requirements of these therapies, logistics providers are leveraging advanced technologies such as:
Internet of Things (IoT): Enables real-time monitoring of temperature and location
Artificial Intelligence (AI): Optimizes delivery routes and predicts potential disruptions
Blockchain: Enhances transparency and traceability across the supply chain
These technologies not only improve operational efficiency but also build trust among stakeholders, including healthcare providers, regulators, and patients.
Key Challenges Facing the Market
Despite its promising growth trajectory, the cell and gene therapy logistics market faces several critical challenges.
1. Packaging and Handling Complexity
CGTs require highly specialized packaging to maintain stability during transit. This includes protection against:
Temperature fluctuations
Physical damage
Contamination
Any failure in packaging can lead to therapy degradation, resulting in significant financial losses and potential risks to patient safety.
2. Extreme Temperature Sensitivity
Many therapies must be stored and transported at ultra-low temperatures, sometimes as low as -150°C. Maintaining these conditions across long distances is a major logistical hurdle.
To address this, companies are investing in:
Temperature-controlled containers
Advanced monitoring systems
Redundant safety measures
Even a brief deviation can compromise the therapy’s effectiveness, making temperature control a non-negotiable requirement.
3. Regulatory Complexity
Transporting biological materials across borders involves navigating a complex web of regulations. Compliance requirements vary by country, adding another layer of complexity to global distribution.
Logistics providers must ensure:
Proper documentation
Adherence to local and international standards
Secure handling of sensitive materials
Regional Insights: A Global Perspective
United States
The U.S. remains a dominant player in the CGT logistics market, driven by a strong biotech ecosystem and high demand for personalized medicine. Logistics providers in the region are focusing on:
Advanced cold chain solutions
Regulatory compliance
Scalable infrastructure
Germany
Germany’s well-established biopharmaceutical sector and strict regulatory framework make it a key market in Europe. The country is witnessing increased demand for logistics services, particularly in biotech hubs like Frankfurt and Düsseldorf.
China
China is rapidly emerging as a major player, fueled by significant investments in biotechnology. The country is leveraging AI and IoT technologies to enhance logistics efficiency and reliability.
Saudi Arabia
Saudi Arabia’s Vision 2030 initiative is driving investments in healthcare infrastructure and biotechnology. The country aims to become a global hub for advanced therapies, creating new opportunities for logistics providers.
Competitive Landscape and Industry Developments
The market is highly competitive, with several key players driving innovation and expansion. Major companies include:
Cencora Corporation
Cardinal Health
McKesson Corporation
EVERSANA
Knipper Health
Arvato SE
DHL
Kuehne+Nagel
Recent developments highlight the industry’s dynamic nature:
In January 2025, Cardinal Health launched Advanced Therapy Connect, a unified ordering platform designed to simplify CGT logistics.
In October 2024, McKesson introduced InspiroGene, a specialized service platform to support the commercialization of cell and gene therapies.
These initiatives underscore the growing importance of integrated, technology-driven logistics solutions.
Market Segmentation Overview
The cell and gene therapy logistics market can be segmented based on several key factors:
By Type
Clinical
Commercial
By Product
Cell Therapies
Gene Therapies
By Therapeutic Area
Oncology
Neurology
Cardiovascular Diseases
Ophthalmology
Infectious Diseases
Others
By End Use
Biopharmaceutical Companies
CDMOs/CMOs
Others
This segmentation reflects the diverse applications and growing demand across multiple healthcare domains.
The Road Ahead: Opportunities and Future Outlook
The future of the cell and gene therapy logistics market is undeniably promising. As personalized medicine continues to gain traction, the need for specialized logistics solutions will only grow.
Key trends shaping the future include:
Expansion of decentralized clinical trials
Increased adoption of digital supply chain technologies
Greater emphasis on sustainability in logistics operations
Development of more robust and scalable cold chain infrastructure
Moreover, as therapies become more accessible, logistics providers will play a crucial role in bridging the gap between innovation and patient care.
Final Thoughts
The rise of cell and gene therapies marks a new era in healthcare—one that prioritizes precision, personalization, and innovation. However, the success of these therapies depends not only on scientific breakthroughs but also on the ability to deliver them safely and efficiently.
The rapid growth of the third-party logistics market reflects this critical need. From advanced cold chain systems to patient-centric delivery models, logistics providers are becoming indispensable partners in the healthcare ecosystem.
As the market moves toward a projected valuation of nearly US$ 21 billion by 2033, it is clear that logistics will remain at the forefront of this transformation—ensuring that groundbreaking therapies reach the patients who need them most, exactly when they need them.






Comments
There are no comments for this story
Be the first to respond and start the conversation.