Isothermal Nucleic Acid Amplification Technology Market Outlook: Point-of-Care Testing, Innovation & Forecast to 2033
How rising demand for rapid clinical diagnostics, point-of-care testing, and molecular detection technologies is driving growth in the isothermal amplification market
Molecular diagnostics has quietly become one of the most consequential corners of healthcare, and isothermal nucleic acid amplification technology — or INAAT — is right at the center of it. Unlike traditional PCR, which requires bulky equipment and precise temperature cycling, INAAT amplifies genetic material at a constant temperature. That single operational difference opens the door to faster results, simpler devices, and testing in places that would otherwise be completely off the table — rural clinics, emergency rooms, food processing plants, even individual homes. The market is expanding rapidly as that practical advantage continues to reach new sectors.
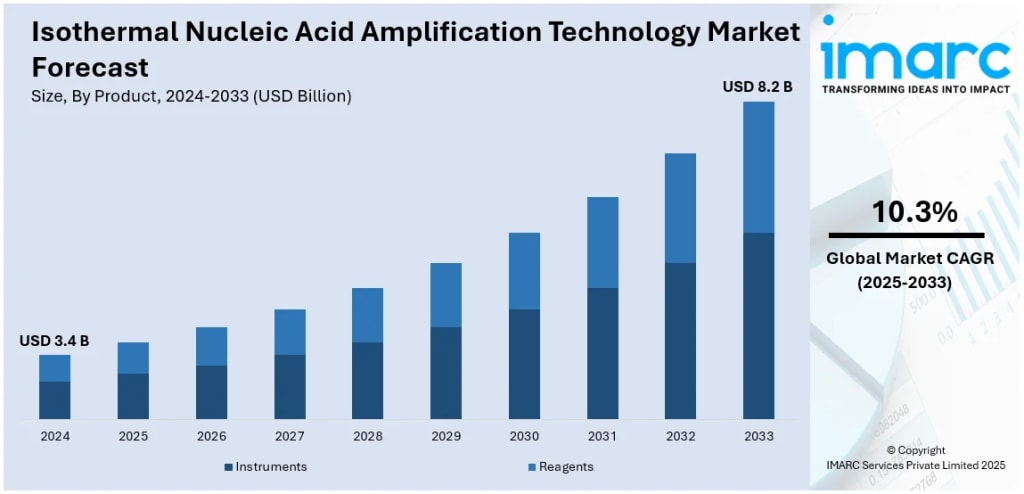
According to IMARC Group's latest research, the global isothermal nucleic acid amplification technology market size was valued at USD 3.4 Billion in 2024. Looking forward, IMARC Group estimates the market to reach USD 8.2 Billion by 2033, exhibiting a CAGR of 10.3% during 2025–2033. North America currently dominates the market, holding a market share of over 43.4% in 2024.
Hospitals are the single largest end-use segment, accounting for roughly 40% of total market activity — driven by the urgent need for fast, reliable diagnostics in emergency care, infectious disease management, and large-scale screening programs. Reagents and kits dominate the product side, while Loop-Mediated Isothermal Amplification (LAMP) leads on the technology front with a 46.4% share, supported by over 500 commercially available LAMP-based diagnostic kits covering pathogens from SARS-CoV-2 to tuberculosis. Infectious disease diagnostics is the primary application segment, representing over 56% of global revenue.
Evaluate Market Potential Before You Invest – Get the Report Sample
Isothermal Nucleic Acid Amplification Technology Market Growth Drivers:
Rising Demand for Fast and Accurate Diagnostics
Speed matters enormously in clinical diagnostics, and INAAT consistently delivers where conventional PCR cannot. Abbott's ID NOW platform — built on isothermal amplification — returns results in under 15 minutes and has been deployed across thousands of urgent care and point-of-care locations in the U.S. For comparison, standard lab-based PCR routinely takes four to six hours when factoring in sample transport and processing. The CDC's Advancing Molecular Epidemiology initiative has specifically funded INAAT-compatible platforms for rapid infectious disease surveillance, recognizing that diagnostic speed translates directly into faster containment decisions and better patient outcomes.
Increasing Prevalence of Infectious Diseases
The global disease burden continues to drive consistent demand for reliable molecular testing. According to the CDC, around 157 million Americans were living with chronic or infectious conditions in recent data, many requiring regular diagnostic monitoring. Influenza, HIV, hepatitis, and tuberculosis together account for tens of millions of annual diagnostic tests in North America alone, and INAAT’s ability to detect pathogen-specific nucleic acids with high sensitivity makes it a natural fit. In low-income settings, where refrigeration and lab infrastructure are limited, isothermal methods are often the only viable molecular diagnostic option, further widening the addressable market globally.
Rapid Advancements in Portable Testing Devices
The miniaturization of INAAT devices has fundamentally changed where and how molecular testing gets done. Smartphone-integrated LAMP setups can now detect pathogens like Zika virus from a single blood drop — a capability that was purely academic just a few years ago. The FDA has granted multiple Emergency Use Authorizations for portable, multi-analyte INAAT systems since 2020, validating both the technology and the commercial pathway. Meridian Bioscience’s Lyo-Ready Direct LAMP saliva mixes are specifically formulated for direct point-of-care use without sample pre-processing — illustrating how product design is increasingly removing technical barriers to adoption in field and home-testing environments.
Expanding Applications Across Healthcare, Food Safety, and Environmental Monitoring
INAAT’s utility goes well beyond hospital diagnostics. In food safety, isothermal amplification platforms are being deployed on production lines to detect bacterial contamination such as Salmonella and Listeria in near real-time — something conventional culture-based testing, which can take days, simply cannot match. Environmental monitoring applications are also gaining ground, with INAAT used in waterborne pathogen detection and biosurveillance. In June 2024, QIAGEN introduced 35 new wet-lab-verified assays for microbial DNA detection, signaling strong commercial confidence in non-clinical application segments. This diversification across sectors is one of the most important long-term growth supports the market has.
Strong Government Initiatives and Institutional Support
Government investment in molecular diagnostics infrastructure has been a consistent tailwind. In November 2024, the U.S. Centers for Disease Control and Prevention formalized a new agreement with industry partners to develop avian influenza diagnostic tests using isothermal amplification approaches — a direct acknowledgment of INAAT’s role in national biosecurity. The EU’s Horizon Europe program has funded multiple molecular diagnostics research consortia, several of which prioritize point-of-care INAAT platforms for low-resource settings. Beyond R&D grants, procurement-side policies in healthcare systems across Asia-Pacific are actively accelerating hospital adoption, particularly in India and China, where government health ministry targets are pushing for expanded diagnostic access in tier-2 and tier-3 cities.
Isothermal Nucleic Acid Amplification Technology Market Trends:
Integration of AI and Digital Health Platforms
AI is increasingly embedded into INAAT workflows, automating result interpretation, flagging anomalies, and integrating outputs directly with electronic health records. This matters practically: reducing reliance on trained technicians lowers the operational cost of point-of-care testing and improves consistency across decentralized settings. Thermo Fisher Scientific launched the CorEvitas clinical registry platform in March 2024, reflecting the broader industry push toward real-world evidence collection at scale. As INAAT devices generate higher test volumes in lower-resource environments, AI-assisted quality control and diagnostic support become less of a feature and more of an operational necessity for maintaining accuracy and regulatory compliance.
Shift Toward Sample-to-Answer Integrated Platforms
The market is moving decisively toward closed, all-in-one diagnostic cartridges that handle sample preparation, amplification, and result readout in a single step. These ‘sample-to-answer’ systems eliminate the most error-prone manual steps in the testing process and make it practical to run molecular diagnostics entirely outside centralized labs. Abbott’s updated ID NOW system, launched in June 2024 for COVID-19, RSV, and Influenza A/B detection, is a leading commercial example. The FDA has cleared multiple integrated INAAT systems under Emergency Use Authorizations, validating both the regulatory appetite and the real-world demand for platforms that make sophisticated molecular diagnostics as operationally simple as a home glucose meter.
Strategic M&A and Partnership Activity Reshaping the Competitive Landscape
The competitive structure of the INAAT market is being actively reshaped through acquisitions and partnerships. In February 2025, Becton, Dickinson and Company announced plans to separate its Biosciences and Diagnostic Solutions unit into a standalone business — a strategic restructuring designed to sharpen focus and accelerate investment in molecular diagnostics. Sherlock Biosciences’ acquisition of Sense Biodetection in 2023 brought together CRISPR-based diagnostics with isothermal nucleic acid amplification capabilities, creating a more powerful combined platform. These moves reflect industry-wide recognition that INAAT is not a niche technology but a foundational infrastructure layer for the next generation of accessible molecular diagnostics globally.
Recent News and Developments in Isothermal Nucleic Acid Amplification Technology Market
• June 2024: Abbott Laboratories released an updated version of its ID NOW™ system for rapid detection of COVID-19, RSV, and Influenza A/B. Using INAAT, the compact and portable device delivers results in under 15 minutes and is specifically designed for urgent care settings and off-site testing locations, expanding point-of-care access for frontline clinicians.
• November 2024: The U.S. Centers for Disease Control and Prevention entered into a new agreement with industry partners to support the development of avian influenza diagnostic tests using isothermal nucleic acid amplification approaches, reflecting growing government reliance on INAAT for national biosurveillance and pandemic preparedness initiatives.
• February 2025: Becton, Dickinson and Company (BD) revealed plans to separate its Biosciences and Diagnostic Solutions business from the rest of the organization. The restructuring is aimed at creating a more focused entity better positioned to invest in and scale molecular diagnostics, including INAAT-based platforms, across global healthcare markets.
• December 2023: Eiken Chemical Co., Ltd. launched an enhanced Loop-mediated Isothermal Amplification (LAMP) kit capable of detecting multiple pathogens simultaneously. The product significantly reduces diagnostic turnaround times in healthcare facilities and addresses the growing clinical need for efficient respiratory infection management.
Note: If you require specific details, data, or insights that are not currently included in the scope of this report, we are happy to accommodate your request. As part of our customization service, we will gather and provide the additional information you need, tailored to your specific requirements. Please let us know your exact needs, and we will ensure the report is updated accordingly to meet your expectations.
About the Creator
Suhaira Yusuf
I specialize in Consumer Insights, focusing on transforming detailed market data into strategic business solutions that accelerate growth and improve customer engagement.




Comments
There are no comments for this story
Be the first to respond and start the conversation.