HPV Testing and PAP Test Market Trends: AI-Based Diagnostics, Digital Cytology & Industry Forecast to 2034
How integration of AI-driven diagnostic tools and digital cytology systems is improving screening outcomes and transforming laboratory workflows in cervical cancer diagnostics
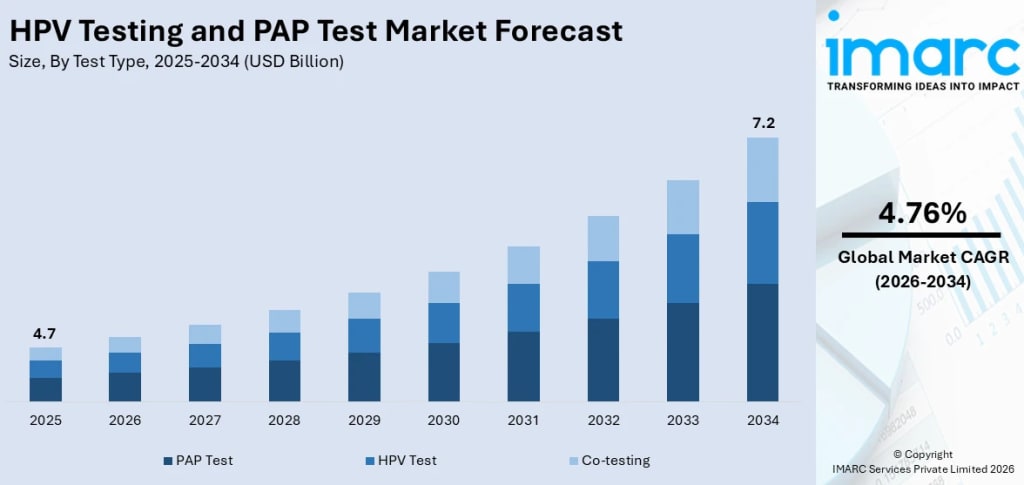
Rising health awareness, advancements in diagnostic technologies, and the increasing burden of cervical cancer are driving the demand for HPV and Pap tests. These efforts are further supported by government-led screening initiatives, the shift toward primary HPV testing, and the introduction of self-sampling kits to improve accessibility. According to IMARC Group’s latest data, The global HPV testing and PAP test market size was valued at USD 4.7 Billion in 2025. Looking forward, IMARC Group estimates the market to reach USD 7.2 Billion by 2034, exhibiting a CAGR of 4.76% from 2026-2034.
The HPV testing and PAP test market has evolved into a critical multi-billion-dollar sector of the global diagnostic industry. Growth is primarily propelled by a transition in clinical guidelines that favor high-sensitivity molecular HPV testing as a primary screening tool over traditional cytology alone. With the World Health Organization (WHO) targeting a 70% screening rate for women globally by 2030, both public and private sectors are scaling up infrastructure. Major segments include consumables such as reagents and collection kits which account for roughly 65% of the market due to their recurring nature. Furthermore, the integration of digital pathology and AI-assisted screening is helping clinical laboratories manage high volumes with greater precision, ensuring that early-stage abnormalities are detected before they progress to invasive malignancies.
HPV Testing and PAP Test Market Growth Drivers:
- Rising Burden of Cervical Cancer and HPV Infections
The increasing global prevalence of human papillomavirus (HPV) infections directly correlates with the rising incidence of cervical cancer, which remains one of the most common cancers in women. Health organizations report nearly 660,000 new cases annually, emphasizing the urgent need for routine screening. In the United States alone, approximately 13,360 new cases of invasive cervical cancer are expected in 2025. This heavy disease burden forces healthcare systems to invest in high-throughput testing platforms. Consequently, the demand for both Pap smears and DNA-based HPV tests continues to grow as clinicians seek to identify high-risk viral strains like HPV 16 and 18 at the earliest possible stage.
- Shift Toward Primary HPV Testing and Updated Clinical Guidelines
Global health authorities are increasingly recommending primary HPV testing as a more sensitive alternative to traditional Pap tests. This transition is a major driver, as molecular tests can detect viral genetic material even before cellular changes are visible under a microscope. For example, the UK National Screening Committee and various European health boards have updated their protocols to favor HPV primary screening, extending the interval between tests for negative results to five years. This policy shift increases the adoption of advanced molecular assays and PCR-based technologies, which now provide higher negative predictive value, giving both patients and healthcare providers greater confidence in long-term screening outcomes.
Get Instant Access to Your Free "HPV Testing and PAP Test Market" Sample Report
- Expansion of Government-Funded Screening and Elimination Initiatives
National health departments are launching aggressive campaigns to meet the WHO’s "90-70-90" elimination targets. Governments are providing significant subsidies and infrastructure support to ensure equitable access to diagnostics. In early 2025, India’s Department of Biotechnology launched indigenous, low-cost HPV kits to support large-scale population screening. Similarly, the UK’s NHS 10-Year Health Plan includes massive investments in diagnostic hubs to reach under-screened populations. These public health schemes provide a steady pipeline of demand for manufacturers, as multi-million-dollar government tenders for consumables and instruments become more frequent, particularly in emerging economies where cervical cancer mortality rates remain high due to previously limited access.
HPV Testing and PAP Test Market Trends:
- Breakthroughs in Self-Sampling and At-Home Collection Kits
A transformative trend is the move toward self-collection kits, which address barriers like patient discomfort, cultural sensitivities, and limited clinic access. In 2026, the UK government is rolling out a major initiative to mail self-sampling HPV kits to women who are overdue for their appointments. Studies indicate that over 50% of physically disabled women and those in remote areas prefer this "DIY" approach. These kits use simple vaginal swabs that are as effective as clinician-collected samples for detecting high-risk HPV. This innovation significantly expands the addressable market by capturing "lost" patients, driving a surge in the consumables segment as millions of single-use collection devices are distributed globally.
- Integration of Artificial Intelligence and Digital Cytology
The diagnostic lab landscape is being reshaped by AI-driven software and automated scanners that assist pathologists in reviewing Pap slides. These AI tools can rapidly flag abnormal cells, reducing the potential for human error and addressing the global shortage of trained cytotechnologists. In 2025, several AI-assisted digital pathology platforms received regulatory clearance, showing an 18-20% improvement in detecting high-grade lesions. This technology allows laboratories to process higher volumes of tests with greater accuracy and lower labor costs. For healthcare providers, this means faster turnaround times and more reliable results, which is essential as screening programs transition to higher-frequency molecular and co-testing models.
- Technological Advancements in Molecular Diagnostics and Genotyping
There is a growing trend toward "extended genotyping," where tests do not just provide a "yes/no" result for HPV but identify the specific viral strain. Advanced platforms, such as Abbott’s Alinity m HR HPV test, which received WHO prequalification in 2025, can detect 14 high-risk genotypes simultaneously. This level of detail allows for personalized risk assessment and better patient management. Additionally, the development of mRNA-based assays is gaining traction because they detect active viral infections rather than just the presence of DNA, reducing false positives. These technical refinements are making screening more precise, helping to avoid unnecessary invasive follow-up procedures like colposcopies for low-risk patients.
Recent News and Developments in HPV Testing and PAP Test Market
- January 2026: NHS England officially began the nationwide rollout of at-home HPV self-sampling kits for individuals overdue for screening by at least six months, aiming to boost participation toward the 80% national target.
- October 2025: A leading diagnostic firm announced a strategic expansion in the Asia-Pacific region, establishing a new high-capacity molecular laboratory in India to support the government's cervical cancer elimination efforts.
- March 2025: The World Health Organization (WHO) prequalified a high-throughput molecular HPV test, facilitating its procurement by international agencies for use in low- and middle-income countries.
Note: If you require specific details, data, or insights that are not currently included in the scope of this report, we are happy to accommodate your request. As part of our customization service, we will gather and provide the additional information you need, tailored to your specific requirements. Please let us know your exact needs, and we will ensure the report is updated accordingly to meet your expectations.
About the Creator
Andrew Sullivan
Hello, I’m Andrew Sullivan. I have over 9+ years of experience as a market research specialist.





Comments
There are no comments for this story
Be the first to respond and start the conversation.